 
Person: Was it trombone? No, Troubador.ΔS° is positive, as expected for a combustion reaction in which one large hydrocarbon molecule is converted to many molecules of gaseous products. The details of how this is done are beyond the level of this text, but Figure 17.4 shows the results for one substance, ammonia. Pg.81 The entropy of a substance, unlike its enthalpy, can be evaluated directly. Entropy of gas at standard conditions (1 bar) T boil: Boiling point: T fus: Fusion (melting) point: d(ln(k H))/d(1/T) Temperature dependence parameter for Henry's Law constant: k H: Henry's Law constant at 298. Yes, cracking a stolen hash is faster, but it's not what the average user should worry about.) The heats of formation, the standard molar entropies at 298 K, are given below. the entropy of a pure substance at 298 K and 1 atm pressure). Standard molar entropies are listed for a reference temperature (like 298 K) and 1 atm pressure (i.e. (Plausible attack on a weak remote web service. Standard Molar Entropy, S 0 The entropy of a substance has an absolute value of 0 entropy at 0 K. (You can add a few more bits to account for the fact that this is only one of a few common formats.) Hf (kJ/mol) Gf (kJ/mol) S (J/mol K) Al (s) 0.0. Source of data: CRC Handbook of Chemistry and Physics,84th Edition (2004). Standard Molar Entropies of Selected Substances at 298 K. Standard thermodynamic Quantities for Chemical Substances at 25C. Uncommon (non-gibberish) base word ]Ĭaps? ]Ĭommon Substitutions ] Table 18.1 Standard Molar Entropies of Selected Substances at 298 K. On each row, the first panel explains the breakdown of a password, the second panel shows how long it would take for a computer to guess, and the third panel provides an example scene showing someone trying to remember the password.)) The comic is laid out with 6 panels arranged in a 3x2 grid. In chemistry, the standard molar entropy is the entropy content of one mole of pure substance at a standard state of pressure and any temperature of interest. A set of boxes is used to indicate how many bits of entropy a section of the password provides. 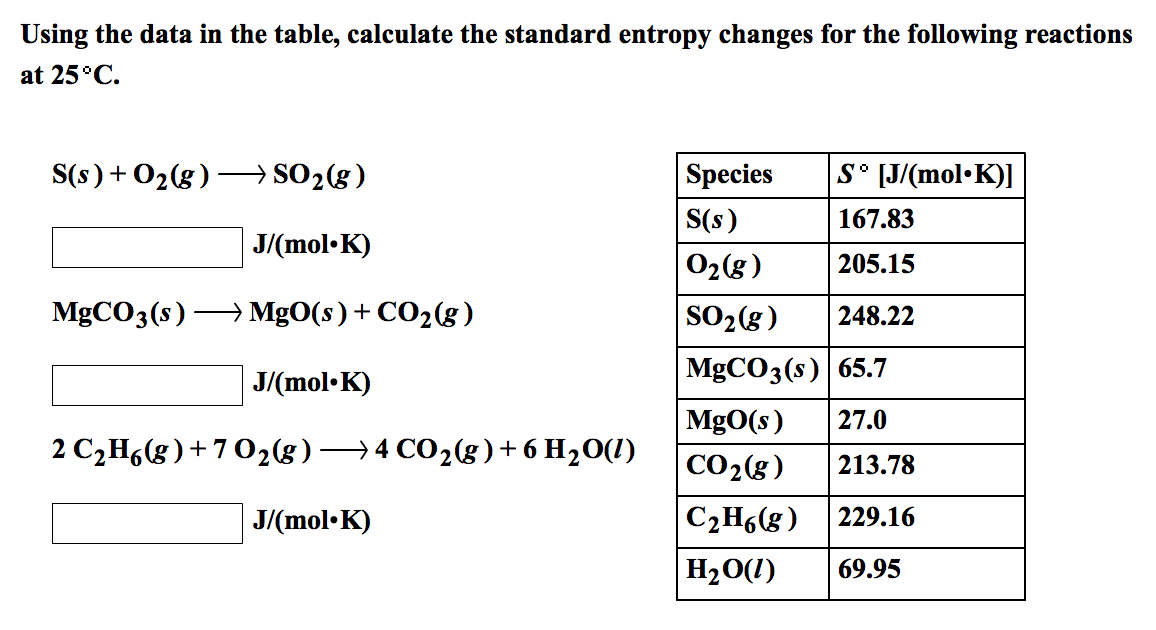
Table l4-4 lists 12 such values, symbolized by S where the superscript denotes the standard state. 2NaCl(s) using the data from the following table: Table showing standard enthalpy, Spontaneity and standard entropy for Na, Cl2 Calculate the standard entropy. When measured at 25 C and 1 atm, these are called standard entropies. SPECIES Hf(298 K) kJ mol1 Hf(298 K) kcal mol1 S(298 K) J K1 mol1 S(298 K) cal K. The entropy decrease for highly polar water from gas > liquid is particularly marked because even in the liquid there are clumps of molecules held together by. Standard entropy, table Chemists have found it possible to assign a numerical quantity for the entropy of each substance. This free textbook is an OpenStax resource written to increase student access to high-quality, peer-reviewed learning materials. As an Amazon Associate we earn from qualifying purchases. Gas-phase entropy and enthalpy values for selected species at 298.15 K and 100 kPa. Thermodynamic data at 25oC for assorted inorganic substances (continued). Values that are calculated or estimated are also indicated in the table. Estimate the value of the standard molar entropy of the straight-chain alkane with the formula. ((The comic illustrates the relative strength of passwords assuming basic knowledge of the system used to generate them. the value of a reference compound selected for this table these are indicated. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
